While COVID-19 vaccinations in the U.S. are continuing apace, calls are growing for a more ambitious strategy to rapidly vaccinate the entire world, and especially in developing nations. Legal proceedings around this issue were triggered in October last year, when India and South Africa circulated a WTO Trade-Related Aspects on IP Rights (TRIPS) petition calling for intellectual property (IP) protections on COVID-19 vaccines to be suspended for the remainder of the pandemic. More than 100 countries signed on to the motion, some out of narrow concern with the pace of vaccine distribution in their country and others as a protest vote against a global IP regime they believe favors rich countries. This year, as we saw second waves in India and elsewhere, the White House tentatively backed a plan to suspend the intellectual property (IP) protections surrounding the vaccines through the World Trade Organization (WTO).
But this has received pushback by many economists and, of course, the pharmaceutical industry, as not necessarily effective, and as counterproductive given the need to incentivize medical R&D in the future. Ultimately, what we need is more manufacturing firepower. The U.S. can best unleash it by coordinating a global technology transfer by purchasing intellectual property and by creating incentives for producers to share their know-how with the rest of the world. Additionally, the U.S. should use its considerable purchasing power to help alleviate supply shortages by paying much more on a per-dose level.
This is an urgent humanitarian, moral, and public health crisis that merits an international response on the scale of the Marshall Plan. The longer the virus hangs around, the higher the risk of a significant mutation that could restart the whole pandemic. In short, we’re not safe from COVID until we are all safe. So let’s tear down any red tape or barriers that may be slowing the production and deployment of vaccines around the world. But how best to do it? Here, I lay out the cases and arguments for how we might get to a Marshall Plan-scale of ambition to help solve the global vaccine shortfall.
The case for suspension of IP
The pro-suspension side is relatively straightforward. While wealthy countries have been able to receive quick access to the vaccine (indeed, near-miraculous progress compared to the normal vaccine creation timeline), deployment has been sluggish around the rest of the world and especially in developing nations:
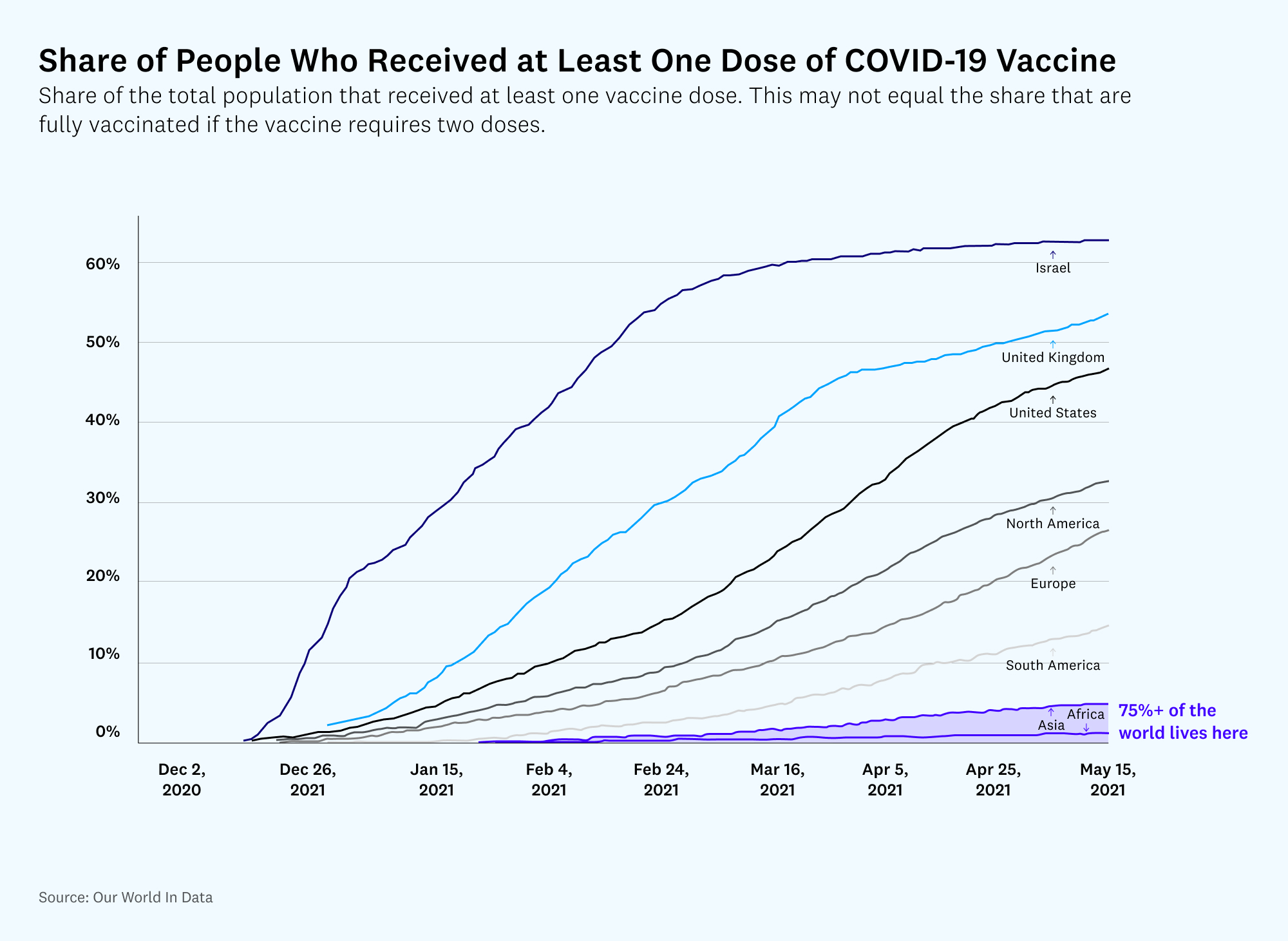
It’s difficult to project vaccine production timelines exactly, but a common estimate from experts seems to be that many low-income countries won’t be fully vaccinated before the end of 2022. Others are more pessimistic, predicting immunity as late as 2024. Either way, that’s still quite a while away, and plenty of time for new COVID variants to emerge and poke holes in our defenses. While we can always create new vaccines or new booster shots to help address new variants, the distribution map is going to be similarly skewed toward rich countries. A cycle in which rich countries continue to get vaccines (and booster shots) first but neglect to accelerate production sufficiently to cover poorer nations in time to prevent mutations is both patently unfair and self-destructive. We are in a race against time, and the costs of accelerating global vaccine production are measured in the billions, whereas the benefits of reaching global immunity a few months earlier are measured in the trillions.
Various organizations and pharmaceutical companies have responded to this situation through the creation of COVAX (for “COVID Vaccine Access”), a pool of pledged vaccines specifically to help vaccinate low-income nations. (It is co-led by the Coalition for Epidemic Preparedness Innovations, the vaccine alliance Gavi, and the World Health Organization). This is an important program and the recent U.S. government investment of $4 billion toward it helps, but the scope is nowhere close to the necessary ambition. The aim of the program is to supply vaccines to help participating counties vaccinate only 20% of their population. COVAX also hit a significant roadblock recently with the news that the Serum Institute of India, meant to be one of the largest producers of COVAX vaccines, won’t be able to resume exports until the end of the year due to the current crisis in India.
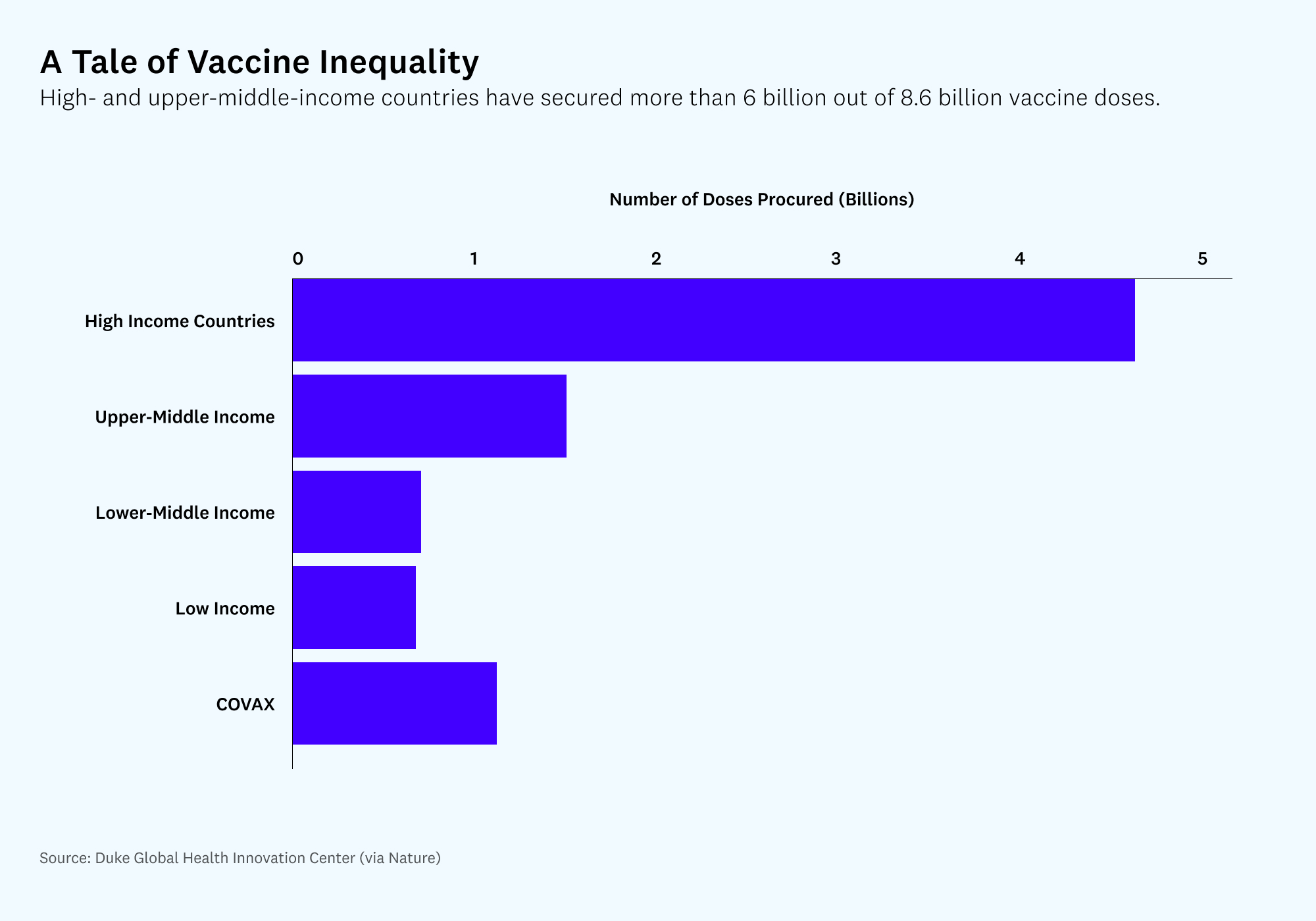
Ultimately, we need to be producing more vaccines in less time. One way of achieving that is to make it easier for competing vaccine manufacturers to get in on the action. Specifically, if there is latent vaccine manufacturing capacity (more on this controversial point later), we could pull out all the stops by suspending the intellectual property protections. This should help speed vaccination efforts around the world, and if the latent manufacturing capacity is located in the Global South, it could make future waves of vaccine booster shots more equitably distributed given the apparent home-market bias of current producers.
The bigger question here is around self-sufficiency and national sovereignty in vaccine and pharmaceutical production.
Much of this conversation is informed by the context of the HIV/AIDS epidemic of the 1990s to early 2000s, which ravaged countries across Africa while the U.S. and other wealthy nations were able to mitigate the damage using antiretroviral treatments. A combination of strict IP enforcement, a lack of foreign aid, and inadequate manufacturing capacity led to millions of deaths that potentially could have been prevented. In 2003, the U.S. created PEPFAR, an extremely successful public health campaign that was able to make massive dents in the crisis by sharing medicine and technical support with partner countries. To counter the concern that IP issues would hamstring future public health responses, the WTO signed the “Doha Declaration”, which created rules around compulsory licensing of medical technology in the event of a public health emergency (but which advocates say is insufficient for this present situation). Given this history, developing nations are understandably skeptical of the idea that they should wait around for Western pharmaceutical companies to produce enough vaccines for them and would like to develop their own manufacturing capacity.
Finally, advocates for suspension typically point out that many of the pharmaceutical companies received R&D funding from the federal government and/or large market commitments through the public-private initiative Operation Warp Speed, which guaranteed them some baseline level of confidence and profitability. Furthermore, the mRNA vaccines themselves are partially the result of decades of public investment in science, and the National Institutes of Health may own some parts of the relevant IP in the case of Moderna.
If you are inclined to believe that pharma R&D is mostly free-riding on the hard work of basic science, then this is a pretty clear-cut case for suspending the IP.
The case against suspension of vaccine IP
On the anti-suspension side, one argument is that these new vaccines (especially the new mRNA vaccines) required billions of dollars and decades of uncertain private investment to reach the technical breakthroughs that have given us a chance to end this pandemic. Basic science work funded by the government helped a lot and certainly established a strong foundation for much of this work. But enterprising individuals with a profit motive played just as large a role. Katalin Karikó, the Hungarian scientist who helped pioneer mRNA vaccines spent most of the 1990s receiving rejection letters for government grants, and ultimately turned to the private sector where she co-founded her own company in 2006.
But even if all the basic research for vaccines were federally funded, the argument for suspension would still create incentive issues. Operationalization and commercialization of scientific breakthroughs are essential, and cost a lot of money. There is a whole series of difficult engineering, logistics, and optimization problems that have to be solved when taking a complex biological product like a vaccine from research to reality. They do not pop out into the world fully formed from a peer-reviewed publication in a scientific journal.
It’s also not an accident that the countries with the most well-developed biotechnology and pharmaceutical clusters are the ones that produced these wonders.
Where most public health interventions failed miserably, the pharmaceutical companies worked around the clock to develop, test, and roll out a whole new genre of vaccine in record-breaking time and at high private cost. Intellectual property helps protect the whole investment pipeline (including the time and cost of failures), making it worth it in the first place; expropriating IP places future R&D investments under a great shadow.
And this is not going to be the last global pandemic we face. We got lucky that COVID-19 has a low fatality rate, relatively speaking, and that we were able to so easily target its spike protein (which is where most of the vaccines concentrated their approaches). On an unlucky Earth Two, these companies spent billions of dollars on a vaccine that flopped and cost them billions of dollars in wasted vaccine development and early preparation.
To ensure that our biology and technology clusters are investing in R&D for new vaccine and therapeutic techniques for the future, we have to align incentives and make it profitable for them to throw billions of dollars at the problem… years before they may ever appear. Furthermore, focusing on intellectual property hides the fact that the formalized information that can be written in a patent application isn’t that useful in isolation. Moderna actually pledged in October that they would not be enforcing the IP rights related to their COVID vaccine. The underlying spike-encoding sequence was already published online by Stanford researchers. Yet no new Moderna knock-offs have sprung up since their October announcement. Why? Because the company has refrained from sharing any details about the manufacturing or design process, which indicates the underlying technical expertise and production process knowledge — the know-how — are just as, if not more, important.
To produce the vaccine, Rachel Silverman from the Center for Global Development observes, manufacturers need access “to the developer’s ‘soft’ IP — the proprietary recipe, cell lines, manufacturing processes and so forth.” Typically this kind of soft IP is transmitted to the new company in a technology transfer process that happens during a licensing deal. Process experts from the licensing firm will sometimes travel to the facilities of the licensee and oversee operations for the first several batches to help convey the sort of tacit knowledge that’s difficult to transmit unless you have physically done the operation yourself. In exchange, the licensing company will get a cut of the revenue from each dose, typically 5-10%. And even when these licensing deals are in place, it’s easy for the new firm to make critical mistakes because the margins for error are razor-thin.
All this means that even if the TRIPS proposal were passed in isolation this month when the WTO meets, it would likely lead to little appreciable change in vaccine production unless paired with significant incentives for the manufacturers to participate in a more formalized technology transfer process.
The open question: Is there latent capacity?
In some sense, the easiest thing to do would be to plow many billions more into programs like COVAX, which reserves vaccine doses for developing nations. That would be good, but it just extends the length of vaccine production rather than increases the rate of production.
Maybe we could specifically help pay for existing vaccine manufacturers to scale up their capacity even more? This, too, would be worthwhile. After all, it’s difficult to imagine having too much vaccine capacity from a social welfare perspective. But even here, building new factories and machines can take months or years, and we need something that works more quickly. This solution also continues to concentrate vaccine production in existing firms and in rich nations, which undermines the self-sufficiency concern that helped fuel the debate in the first place.
The main advantage that some version of IP suspension offers is activating manufacturing capacity that already exists but that isn’t currently being deployed. Just how much latent capacity is there?
For the mRNA vaccines of Pfizer and Moderna, it appears that production bottlenecks are quite severe. A great piece by Derek Lowe outlines how specialized the supply chains and manufacturing processes for mRNA vaccines are, especially in their use of lipid nanoparticles. Lowe argues that only a handful of firms globally could have jumped in and started making mRNA vaccines immediately, and it seems likely that all of them already have.
However, this may be too fatalistic about our ability to bring new manufacturers on board over a slightly longer time horizon. Production bottlenecks will ease over time with enough investment and in learning by doing. After all, no one was manufacturing mRNA vaccines before the crisis, but we’ve obviously been able to create new capacity over time. And there’s enormous investment happening now to expand production of scarce raw materials. We’re seeing supply-chain maturity by fire.
It seems more plausible, however, that there is latent capacity using the more traditional adenoviral vector vaccines like Johnson & Johnson and AstraZeneca, or the protein subunit vaccines like Novavax. Because these older vaccine techniques have the advantage of having existed for longer, more vaccine manufacturers around the world have the capacity to produce them than can produce mRNA vaccines right now. The related supply chains are more mature as well, which means a sudden surge in demand can be better accommodated.
Meanwhile, ongoing and recent voluntary licensing deals seem to be missing willing manufacturers who want to get in on the action. The Canadian firm Biolyse Pharma, for example, has capacity to make around 50 million vaccines a year, and has spent months trying to arrange a license — but has received no offers. Based on the kind of machinery they already have in-house, they believe they are capable of manufacturing the Johnson & Johnson vaccine in as few as 3-6 months if they had access to the underlying IP. In an unusual step, they’ve already made a contract with the government of Bolivia to produce at least 15 million vaccines in the event that the WTO waiver goes into effect.
The managing director of Bangladesh-based Incepta Pharmaceuticals has claimed they have the capacity to create 350 million protein subunit vaccines, like those used by Novavax, as well as the capacity to fill-and-finish 500 million vaccine doses (taking the bulk vaccine containers and transferring them to final vials). As the company’s chairman told the Wall Street Journal, “We have a tremendous facility here lying idle. It is very frustrating.” They are willing to pay IP fees, but haven’t received a response from U.S. manufacturers. Similarly, Teva Pharmaceutical Industries in Israel recently announced that they’ve stopped searching for a vaccine partnership because all the primary manufacturers were essentially booked up.
What these examples, and others like them, show is that there is physical manufacturing capacity that could plausibly be activated (particularly with adenoviral or subunit vaccines). But that willingness and/or ability by primary vaccine makers to take on new voluntary licensing agreements with outside partners is the bottleneck.
There may also be capacity that is not exactly latent, but is not being used very efficiently. If an opportunity presented itself, we could see some vaccine manufacturers who are currently pursuing vaccine trials drop their research trials and focus on producing known (and already approved) vaccines. Sanofi and GlaxoSmithKline, for example, began a phase 2 clinical trial in February that (if everything goes right) will finally be ready in Q4 of this year. There’s no particular reason to think this new vaccine will be better than the existing offerings. It’s more likely that they are pursuing this line because they want to have proprietary IP to offer. While both Sanofi and GSK have signed deals to help manufacture other vaccines in the meantime, these are quite limited and the companies are likely saving capacity so they can rapidly retool if their own trials are promising. But it would certainly be more socially valuable if we could encourage them to drop these trials and fully ramp up the production of proven vaccines right now.
The case for patent buyouts
The economist Michael Kremer wrote a paper in 1997 formalizing the idea of using patent buyouts as a way of maintaining strong incentives for innovation, while still getting crucial information into the public domain as soon as possible. Essentially, a government could offer to pay the present value of the expected future revenue stream that would result from the temporary monopoly that a patent sometimes grants. While the patent-owning company or individual should be indifferent to the outcome, the general public could receive more value from having unabridged access to the information and the ability to modify it without permission before the patent expired. In these cases, a patent buyout can clearly improve outcomes for everyone, and Kremer notes that pharmaceuticals may be a particularly appropriate case.
Patent buyouts maximize the number of players that can legally produce vaccines while maintaining strong incentives for future innovation. Of course, in this situation it’s only partly about the intellectual property, and partly about the manufacturing know-how that has to be transferred, which means we need a broader conception here — a more full-stack “technology buyout” that includes both the IP and the process knowledge transfer.
Essentially, the U.S. government (or even a set of governments) could offer a lump sum payment to the accepting firm(s) to make explicit the scientific and production process as much as possible and then also make it publicly available. We could offer a secondary payment for sharing on-the-ground technical expertise to aid in setting up manufacturing operations — either on an individual factory level, or on the basis of vaccine doses administered. A per-vaccine-dose-administered basis properly aligns incentives for the firm(s) sharing technology, maximizing their impact by transferring to partners that can actually get shots into arms as quickly as possible and to make sure they do a good job.
To put some back-of-the-envelope numbers on this, the initial lump sum payment to make the IP public would be in the range of $10-20 billion per firm, and the additional per-dose-administered prize would be in the realm of $0.50-$2. Assuming this program was able to administer vaccines for an additional 4 billion people (8 billion doses) across the developing world, we are talking in the range of $36-56 billion.
And we should overpay. In a situation like this, we should err on the side of overcompensating, and risking some economic rents, rather than inadvertently undercompensating and hurting the long-term incentives for innovation. The key is to not kill the goose that lays the golden egg. In any event, we should be willing to pay an order of magnitude more than $36 billion to definitively end COVID, so this program is a bargain under a wide range of potential cost assumptions. One estimate from a group of economists and public health officials ballpark the global monthly cost of the pandemic at around $1 trillion per month.
Payments for each vaccine dose administered may sound very similar to the voluntary licensing agreements that manufacturers around the world have already signed. It is a closely related mechanism; commentators like Rachel Silverman (quoted above) have actually suggested that the best way forward may be having the federal government use its political leverage to lean on U.S. pharmaceutical companies to accept more licensing deals with manufacturers in developing countries. Which raises the question, why bother doing the additional lump sum buyout to make the information public? Shouldn’t we concentrate all our efforts on encouraging licensing?
I would differentiate the technology buyout I’m suggesting from voluntary licensing on a few dimensions:
- First, buying out the IP may be a determining factor for encouraging large, mature manufacturers like GSK and Sanofi to abandon their own duplicative vaccine trials and go full steam toward producing existing vaccines as it removes any competitive disadvantage in paying the licensing fee.
- Second, because these voluntary licensing deals are typically assigned as a percentage of the dose cost, it creates an incentive for licensing firms to prioritize deals that will charge a higher price to the buying local governments where that may not be optimal. If the U.S. government is instead paying for each dose administered in these developing nations, then the transferring firm(s) should be neutral with regard to the sale price.
- Third, this structure allows the federal government to selectively overpay on a per-dose basis if the U.S. firm is genuinely helping a developing nation jumpstart new manufacturing capacity as opposed to helping an existing manufacturer retool.
- Fourth, individual countries and foreign manufacturers know their own capacity better than U.S. government officials or even U.S. firms do, so opening up the IP could help them identify latent manufacturing opportunities more effectively than a top-down approach.
- Fifth, opening up the IP at least gives every nation the ability to try and make their own vaccine if they so wish, which helps address self-sufficiency concerns.
A buyout also presents several key advantages when compared to IP suspension as well, even putting the incentive issues aside:
- First, speed. Even with the U.S. reversing course and supporting the WTO proposal, it will take quite a bit of time to negotiate and wrangle all the other countries to the table. Remember, the proposal has to be supported unanimously, so there is no guarantee that all other opposed nations will reverse course just because the U.S. did. A buyout, in contrast, can be done unilaterally by the U.S. (at least for the U.S.-based vaccine firms).
- Second, in the unfortunate event that a new variant requires a booster shot, a buyout ensures the incentive to quickly create a solution so that the new booster shot can also get bought out. Under the WTO petition, the IP suspension would remain in play for the duration of the crisis, which would reduce the urgency and resources pharma companies are willing to throw at the problem, given fewer opportunities to recoup their costs.
While a technology buyout would help accelerate the sharing of technical know-how with the world, it does not necessarily address the supply chain bottlenecks of critical ingredients. These shortages range from ingredients as mundane as plastic bags to as exotic as Chilean tree bark. To help address these shortages, the single best thing the U.S. could do is offer to pay much higher prices for each vaccine dose produced, as this would allow the higher price signal to work its way through the supply chain and give manufacturers more fiscal capacity to pay higher prices to suppliers for ingredients.
Building for the long term
There is the issue of whether a technology buyout would be better suited for the mRNA vaccines of Modena and Pfizer or for a more traditional vaccine like Johnson & Johnson. To answer that we have to ask a more fundamental question: What is it we are trying to achieve here? There is likely more short-term capacity to scale adenoviral vector vaccines, so if we are narrowly trying to get the world vaccinated against COVID-19 as quickly as humanly possible, then a technology buyout for the J&J vaccine probably makes the most sense.
But if we have a larger vision of using this crisis as an opportunity to bootstrap new, flexible vaccine manufacturing capacity around the world for the future, then mRNA vaccines have a host of advantages that make them the better long-term pick. (Of course, we can try to do both to take advantage of the distinct advantages of each vaccine type, but it helps to have a longer term anchor goal to build toward.) I would argue a few points in favor of the future-oriented approach:
- First, hoping the adenoviral vector vaccines hold up long enough against possible variants is a significant risk. As alluded to earlier, the mRNA vaccines appear to be more resilient to the recent COVID variants and are significantly easier to modify in the event a new variant arrives that our current vaccines can’t handle.
- Second, mRNA vaccines could unlock a much broader wave of medical improvements, including possible vaccines for multiple sclerosis, some forms of cancer, malaria, and HIV. All of which means mRNA is likely to be a more general-purpose vaccine technology and makes it almost impossible to imagine having overbuilt capacity at this point.
- Third, separate from the supply chain issues, new mRNA facilities are actually much cheaper to build and operate than traditional vaccine factories. From an article in the Journal of Advanced Manufacturing and Processing: “Based on our techno-economic assessment, the RNA vaccine production process can be two to three orders of magnitude smaller than conventional vaccine production processes in terms of facility scale, and can be constructed in less than half the time with 1/20 to 1/35 of the upfront capital investment… Due to its small scale, the RNA vaccine drug substance production process could be placed in a small part of an existing conventional vaccine facility, for example in a room, and still produce more doses worth of drug substance than the entire original conventional vaccine production facility.”
- Fourth, we should view this as an opportunity to build goodwill around the developing world. The total U.S. foreign aid budget was around $40 billion in 2019, right in the range of what we are proposing here. I’m inclined to believe sharing new, highly effective technology around the world during this unique crisis would generate a significantly higher return diplomatically than the projects we usually get with this scale of funds. Already we’ve seen China and Russia attempt to leverage their vaccine exports for diplomatic purposes. Much like the Belt and Road initiative that has helped China make inroads across Africa, the U.S. would be helping with technological infrastructure investment.
Finally, it is in the long-term interest of all of humanity to have a developed and coordinated ability to respond to new diseases around the world. Viruses don’t respect national borders and the risk of a global pandemic has only increased over time as our world has become more interconnected. This capacity inherently needs to be distributed around the world for it to be maximally effective, so we may as well start building it out now. The capacity to rapidly create and distribute new mRNA vaccines could help us eliminate much of the long-term risk of bioweapons and natural pandemics. That is the true goal worth aspiring toward here.
* * *
The U.S. should be aiming not only to vaccinate the entire world, but to teach the world how to make vaccines. As the saying goes: Give the world vaccines and you stop one pandemic; teach the world how to manufacture vaccines and you stop pandemics forever.
Views expressed in “posts” (including articles, podcasts, videos, and social media) are those of the individuals quoted therein and are not necessarily the views of AH Capital Management, L.L.C. (“a16z”) or its respective affiliates. Certain information contained in here has been obtained from third-party sources, including from portfolio companies of funds managed by a16z. While taken from sources believed to be reliable, a16z has not independently verified such information and makes no representations about the enduring accuracy of the information or its appropriateness for a given situation.
This content is provided for informational purposes only, and should not be relied upon as legal, business, investment, or tax advice. You should consult your own advisers as to those matters. References to any securities or digital assets are for illustrative purposes only, and do not constitute an investment recommendation or offer to provide investment advisory services. Furthermore, this content is not directed at nor intended for use by any investors or prospective investors, and may not under any circumstances be relied upon when making a decision to invest in any fund managed by a16z. (An offering to invest in an a16z fund will be made only by the private placement memorandum, subscription agreement, and other relevant documentation of any such fund and should be read in their entirety.) Any investments or portfolio companies mentioned, referred to, or described are not representative of all investments in vehicles managed by a16z, and there can be no assurance that the investments will be profitable or that other investments made in the future will have similar characteristics or results. A list of investments made by funds managed by Andreessen Horowitz (excluding investments for which the issuer has not provided permission for a16z to disclose publicly as well as unannounced investments in publicly traded digital assets) is available at https://a16z.com/investments/.
Charts and graphs provided within are for informational purposes solely and should not be relied upon when making any investment decision. Past performance is not indicative of future results. The content speaks only as of the date indicated. Any projections, estimates, forecasts, targets, prospects, and/or opinions expressed in these materials are subject to change without notice and may differ or be contrary to opinions expressed by others. Please see https://a16z.com/disclosures for additional important information.
